Chemical identification, antioxidant, cholinesterase inhibitory, and cytotoxic properties of fucoidan extracted from Persian Gulf Sargassum angustifolium
-
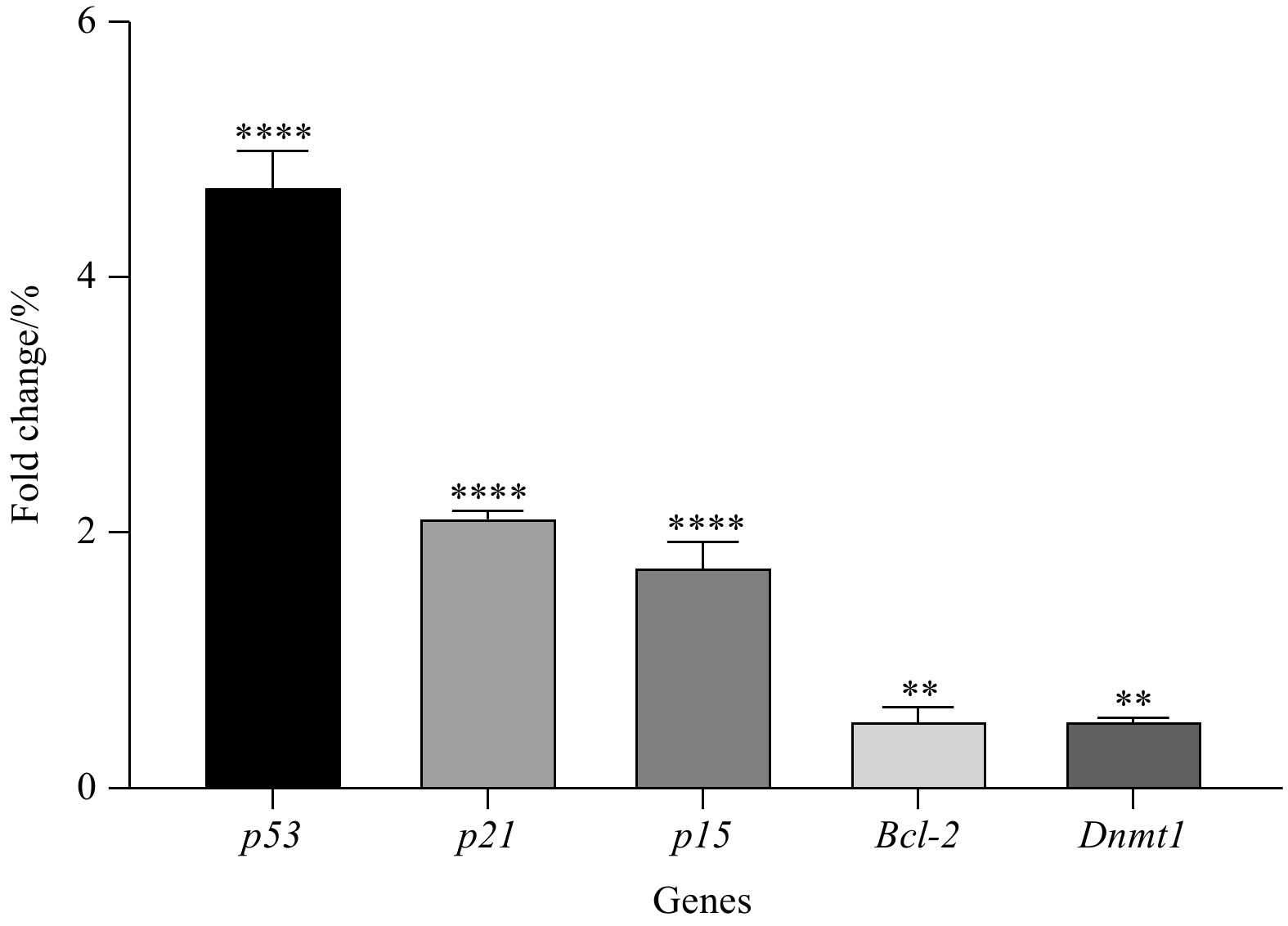
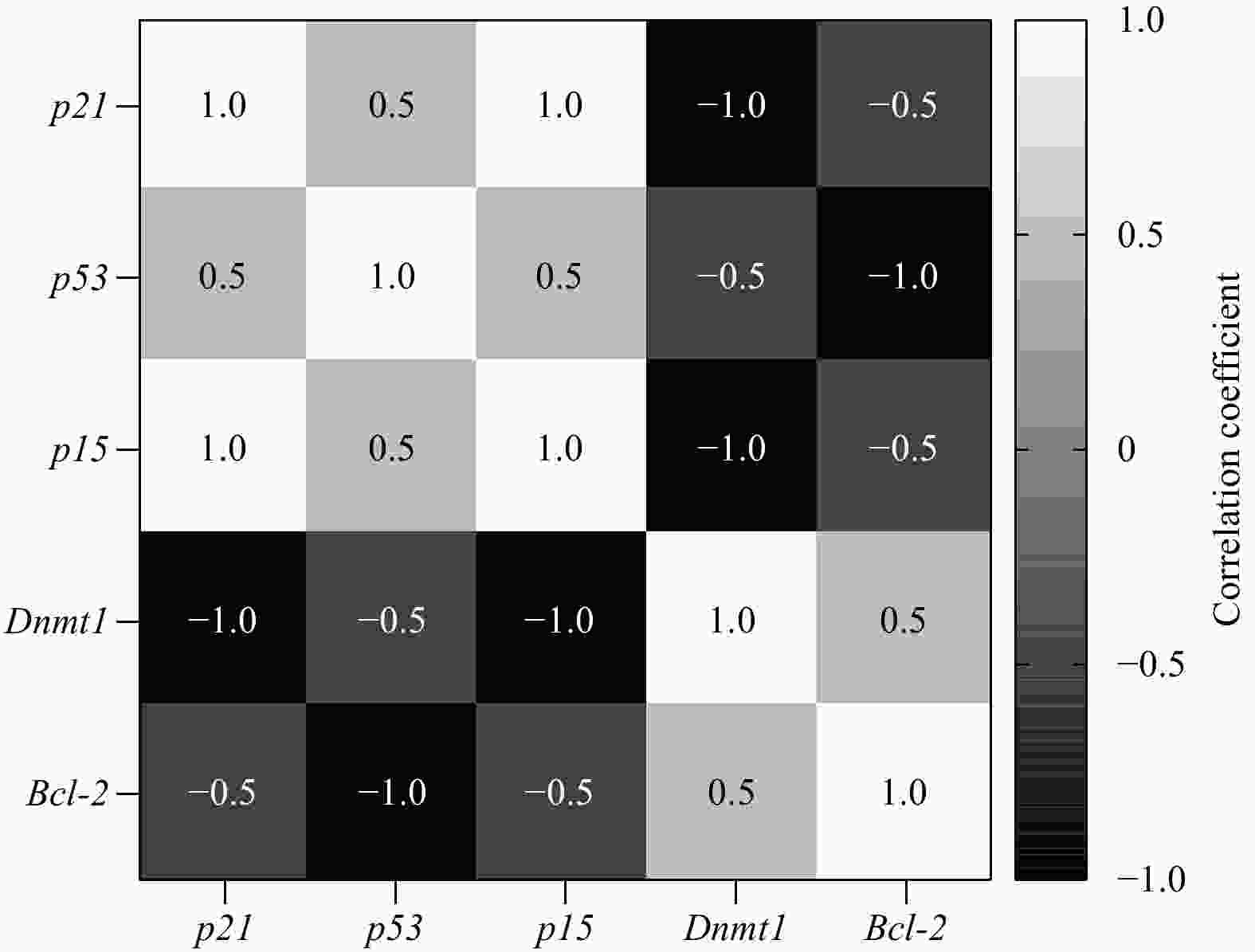
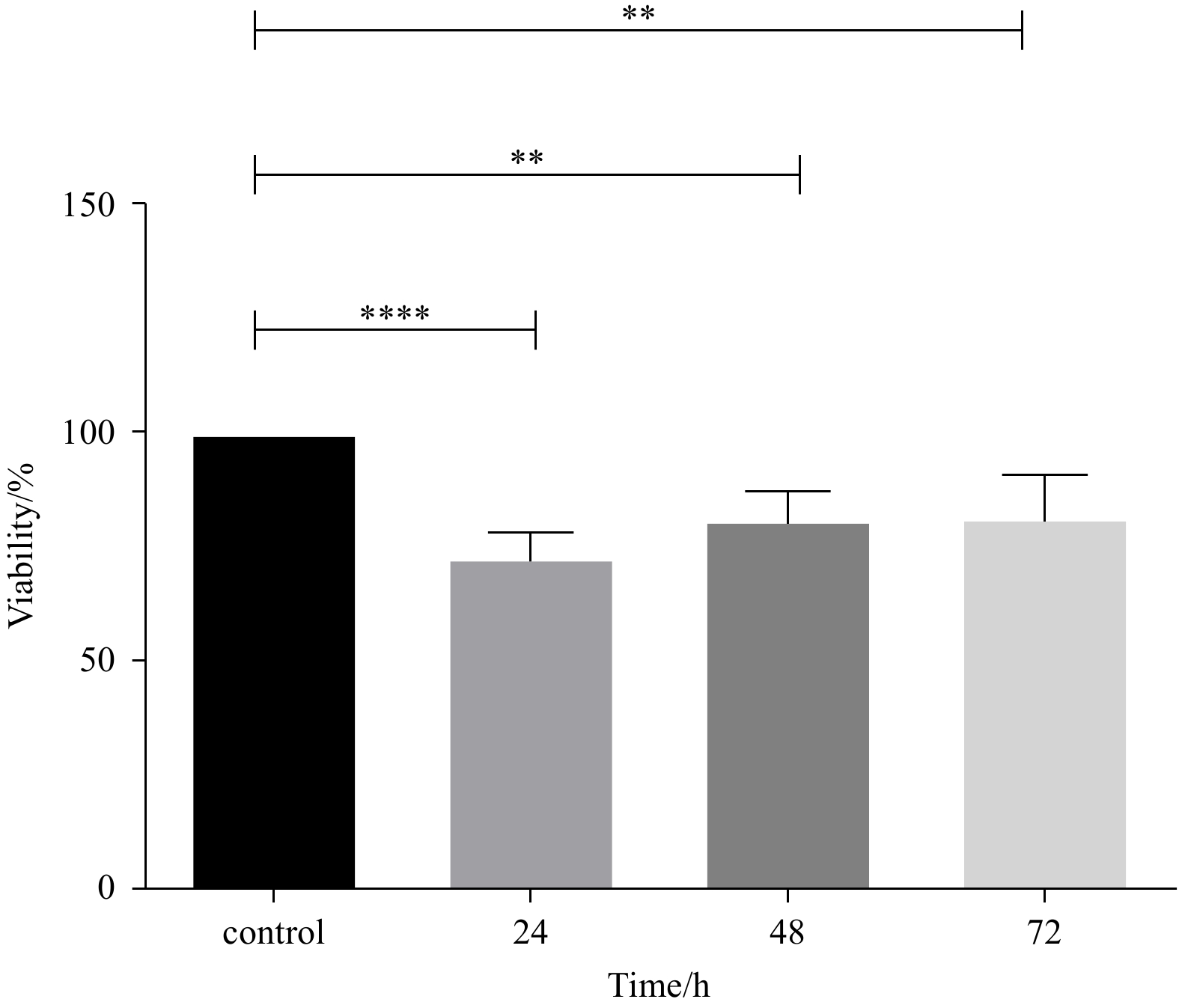
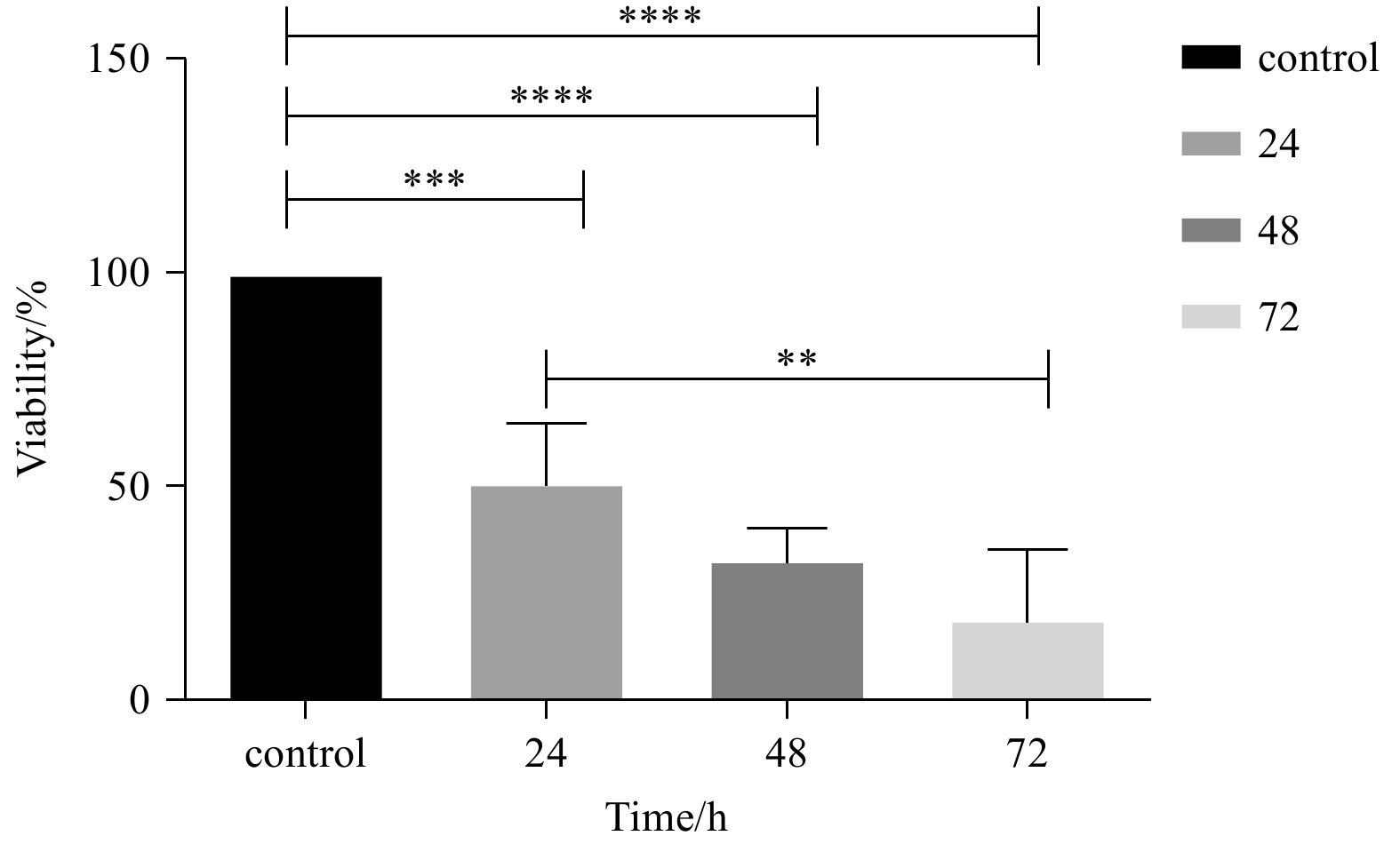
Abstract: Marine macroalgal sulfated fucose-containing polysaccharides, like fucoidan, have drawn significant attention due to their biotechnological potentials, such as anti-cancer, antioxidant, and anti-cholinesterase activities. The fucoidan derived from brown macroalgae Sargassum angustifolium species (FSA) was investigated for its cytotoxic effects and alterations in cell proliferation, and cell cycle-related gene expression in the present study occurred on NB4 cell line. The results showed that FSA would induce p53, p21, pro-apoptotic genes and increase expression of the p15 gene as a cell arrest marker. Also, FSA inhibited the anti-apoptotic effect of the Bcl-2 gene and decreased dnmt-1 gene expression. FSA significantly exhibited potent 2, 2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging activity (p<0.05) with an IC50 value of 0.157 mg/mL and showed moderate anti-acetylcholinesterase activity with an IC50 value of 1.20 μg/mL. These results indicated the potential of FSA for the development of therapeutic or preventive agents of cancer and Alzheimer’s disease mainly through cytotoxic effect and AChE (acetylcholinesterase) inhibition as well as additional antioxidant capacities.
-
Key words:
- fucoidan /
- Sargassum angustifolium /
- cytotoxicity /
- antioxidant /
- anti-cholinesterase
-
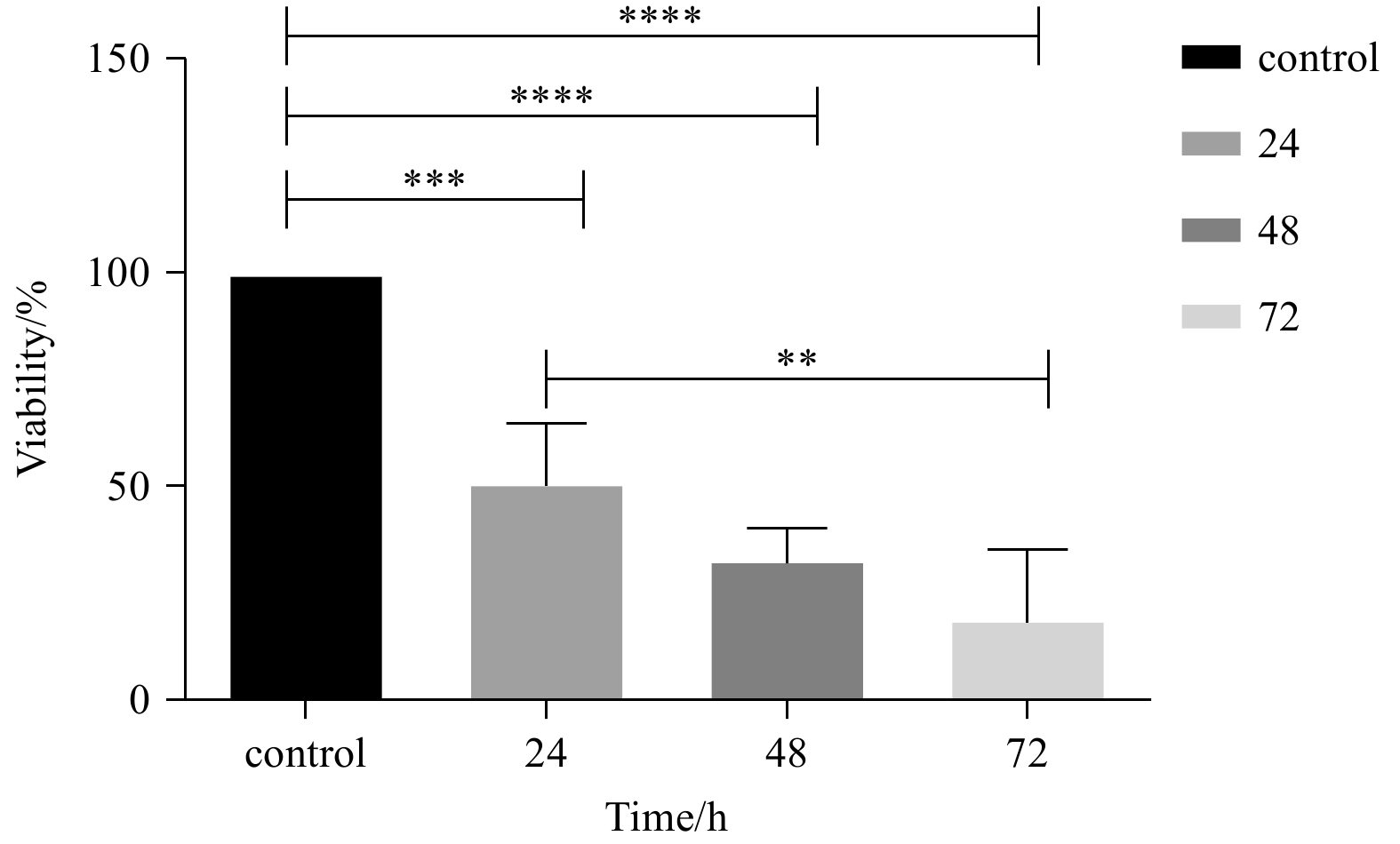
Figure 6. Diagram of the anti-cholinesterase activity of fucoidan extracted from sargassum angustifolium. The anti-cholinesterase activity of fucoidan in concentrations of 0.01 μg/mL, 0.1 μg/mL, 1 μg/mL, 10 μg/mL, and 100 μg/mL compared with the control group (10% DMSO in buffer). The values are presented as mean±SEM of 3 independent experiments (n=3) and analyzed by one-way analysis of variance (ANOVA) (*p<0.05, **p<0.01 and ***p<0.001 compared with the control group).
Table 1. Sequences of primers used in qRT-PCR
Base pair Forward (5'–3') Reverse (5'–3') Gene 111 TGGACAGGACTGAACGTCTTG CCAGCAGGTCAGCAAAGAATTTA HPRT 90 CGGTGGGGTCATTTGTGTG CGGTTCAGGTACTCAGTCATCC Bcl-2 118 ACTCATCCGATTTGGCTCTTTC CCTAGCCCCAGGATTACAAG Dnmt1 82 GATGTGCAAGCGACGACAGA GAGCAAAGGCCAGCATCCT p15 130 CCTCTCACTGTCTTGTACCCT GCGTTTGGAGTGGTAGAAATCT p21 121 TAACAGTTCCTGCATGGGCGGC AGGACAGGCACAAACACGCACC p53 Table 2. Sugar analysis of the polysaccharide from Sargassum angustifolium
Sugar Composition/% Fucose 53±2.0 Galactose 0.2±0.9 Mannose 0.3±0.7 Glucose 12.6±0.4 Uronic acids 0.36±0.5 Note: Each value represents the percentage of monosaccharides obtained in the fraction. -
Adams J, Nassiri M. 2015. Acute promyelocytic leukemia: a review and discussion of variant translocations. Archives of Pathology & Laboratory Medicine, 139(10): 1308–1313 Adhami H R, Farsam H, Krenn L. 2011. Screening of medicinal plants from Iranian traditional medicine for acetylcholinesterase inhibition. Phytotherapy Research, 25(8): 1148–1152. doi: 10.1002/ptr.3409 Ahmady-Asbchin S, Mohammadi M. 2011. Biosorption of copper ions by marine brown alga Fucus vesiculosus. Journal of Biological and Environmental Sciences, 5(15): 121–127 Atashrazm F, Lowenthal R M, Woods G M, et al. 2015. Fucoidan and cancer: a multifunctional molecule with anti-tumor potential. Marine Drugs, 13(4): 2327–2346. doi: 10.3390/md13042327 Bahramzadeh S, Tabarsa M, You S G, et al. 2019. Purification, structural analysis and mechanism of murine macrophage cell activation by sulfated polysaccharides from Cystoseira indica. Carbohydrate Polymers, 205: 261–270. doi: 10.1016/j.carbpol.2018.10.022 Barbui T, Finazzi G, Falanga A. 1998. The impact of all-trans-retinoic acid on the coagulopathy of acute promyelocytic leukemia. Blood, 91(9): 3093–3102. doi: 10.1182/blood.V91.9.3093 Benslima A, Sellimi S, Hamdi M, et al. 2021. Brown seaweed Cystoseira schiffneri as a promising source of sulfated fucans: seasonal variability of structural, chemical, and antioxidant properties. Food Science & Nutrition, 9(3): 1551–1563 Bernard J, Weil M, Boiron M, et al. 1973. Acute promyelocytic leukemia: results of treatment by daunorubicin. Blood, 41(4): 489–496. doi: 10.1182/blood.V41.4.489.489 Bolognesi M L, Matera R, Minarini A, et al. 2009. Alzheimer’s disease: new approaches to drug discovery. Current Opinion in Chemical Biology, 13(3): 303–308. doi: 10.1016/j.cbpa.2009.04.619 Boo H J, Hong J Y, Kim S C, et al. 2013. The anticancer effect of fucoidan in PC-3 prostate cancer cells. Marine Drugs, 11(8): 2982–2999. doi: 10.3390/md11082982 Borazjani N J, Tabarsa M, You S G, et al. 2018. Purification, molecular properties, structural characterization, and immunomodulatory activities of water soluble polysaccharides from Sargassum angustifolium. International Journal of Biological Macromolecules, 109: 793–802. doi: 10.1016/j.ijbiomac.2017.11.059 Carpinella M C, Andrione D G, Ruiz G, et al. 2010. Screening for acetylcholinesterase inhibitory activity in plant extracts from Argentina. Phytotherapy Research, 24(2): 259–263. doi: 10.1002/ptr.2923 Castaigne S, Chomienne C, Daniel M T, et al. 1990. All-trans retinoic acid as a differentiation therapy for acute promyelocytic leukemia. I. clinical results. Blood, 76(9): 1704–1709. doi: 10.1182/blood.V76.9.1704.1704 Chen Zixing, Xue Yongquan, Zhang Ri, et al. 1991. A clinical and experimental study on all-trans retinoic acid-treated acute promyelocytic leukemia patients. Blood, 78(6): 1413–1419. doi: 10.1182/blood.V78.6.1413.1413 Choi B W, Ryu G, Park S H, et al. 2007. Anticholinesterase activity of plastoquinones from Sargassum sagamianum: lead compounds for Alzheimer’s disease therapy. Phytotherapy Research, 21(5): 423–426. doi: 10.1002/ptr.2090 Choo G S, Lee H N, Shin S A, et al. 2016. Anticancer effect of fucoidan on DU-145 prostate cancer cells through inhibition of PI3K/Akt and MAPK pathway expression. Marine Drugs, 14(7): 126. doi: 10.3390/md14070126 Degos L, Chomienne C, Daniel M T, et al. 1990. Treatment of first relapse in acute promyelocytic leukaemia with all-trans retinoic acid. Lancet, 336(8728): 1440–1441 Devi L B, Das S K, Mandal A B. 2014. Impact of surface functionalization of AgNPs on binding and conformational change of hemoglobin (Hb) and hemolytic behavior. The Journal of Physical Chemistry C, 118(51): 29739–29749. doi: 10.1021/jp5075048 Ellman G L, Courtney K D, Andres Jr V, et al. 1961. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochemical Pharmacology, 7(2): 88–90, IN1, 91–95 Farvin K H S, Jacobsen C. 2013. Phenolic compounds and antioxidant activities of selected species of seaweeds from Danish coast. Food Chemistry, 138(2–3): 1670–1681 Fenaux P, Chastang C, Chevret S, et al. 1999. A randomized comparison of all transretinoic acid (ATRA) followed by chemotherapy and ATRA plus chemotherapy and the role of maintenance therapy in newly diagnosed acute promyelocytic leukemia. Blood, 94(4): 1192–2000. doi: 10.1182/blood.V94.4.1192 Fitton J H, Stringer D N, Karpiniec S S. 2015. Therapies from fucoidan: an update. Marine Drugs, 13(9): 5920–5946. doi: 10.3390/md13095920 Frankel S R, Eardley A, Lauwers G, et al. 1992. The “retinoic acid syndrome” in acute promyelocytic leukemia. Annals of Internal Medicine, 117(4): 292–296. doi: 10.7326/0003-4819-117-4-292 Gao Yonglin, Li Chunmei, Yin Jungang, et al. 2012. Fucoidan, a sulfated polysaccharide from brown algae, improves cognitive impairment induced by infusion of Aβ peptide in rats. Environmental Toxicology and Pharmacology, 33(2): 304–311. doi: 10.1016/j.etap.2011.12.022 Grimwade D, Biondi A, Mozziconacci M J, et al. 2000. Characterization of acute promyelocytic leukemia cases lacking the classic t (15; 17): results of the European Working Party. Blood, 96(4): 1297–1308 Harvey A L. 2008. Natural products in drug discovery. Drug Discovery Today, 13(19–20): 894–901 Hodges J R. 2006. Alzheimer’s centennial legacy: origins, landmarks and the current status of knowledge concerning cognitive aspects. Brain, 129(11): 2811–2822. doi: 10.1093/brain/awl275 Hu Pei, Li Zhixiong, Chen Mingcang, et al. 2016. Structural elucidation and protective role of a polysaccharide from Sargassum fusiforme on ameliorating learning and memory deficiencies in mice. Carbohydrate Polymers, 139: 150–158. doi: 10.1016/j.carbpol.2015.12.019 Irhimeh M R, Fitton J H, Lowenthal R M. 2007. Fucoidan ingestion increases the expression of CXCR4 on human CD34+ cells. Experimental Hematology, 35(6): 989–994. doi: 10.1016/j.exphem.2007.02.009 Jenny M, Klieber M, Zaknun D, et al. 2011. In vitro testing for anti-inflammatory properties of compounds employing peripheral blood mononuclear cells freshly isolated from healthy donors. Inflammation Research, 60(2): 127–135. doi: 10.1007/s00011-010-0244-y Jin J O, Song M G, Kim Y N, et al. 2010. The mechanism of fucoidan-induced apoptosis in leukemic cells: involvement of ERK1/2, JNK, glutathione, and nitric oxide. Molecular Carcinogenesis, 49(8): 771–782 Kakizuka A, Miller Jr W H, Umesono K, et al. 1991. Chromosomal translocation t (1.5; 17) in human acute promyelocytic leukemia fuses RARα with a novel putative transcription factor, PML. Cell, 66(4): 663–674. doi: 10.1016/0092-8674(91)90112-C Kandasamy S, Khan W, Kulshreshtha G, et al. 2015. The fucose containing polymer (FCP) rich fraction of Ascophyllum nodosum (L. ) Le Jol. protects Caenorhabditis elegans against Pseudomonas aeruginosa by triggering innate immune signaling pathways and suppression of pathogen virulence factors. Algae, 30(2): 147–161 Lindsley R C, Mar B G, Mazzola E, et al. 2015. Acute myeloid leukemia ontogeny is defined by distinct somatic mutations. Blood, 125(9): 1367–1376. doi: 10.1182/blood-2014-11-610543 Martins A, Vieira H, Gaspar H, et al. 2014. Marketed marine natural products in the pharmaceutical and cosmeceutical industries: tips for success. Marine Drugs, 12(2): 1066–1101. doi: 10.3390/md12021066 Molinski T F, Dalisay D S, Lievens S L, et al. 2009. Drug development from marine natural products. Nature Reviews Drug Discovery, 8(1): 69–85. doi: 10.1038/nrd2487 Mukherjee P K, Kumar V, Houghton P J. 2007. Screening of Indian medicinal plants for acetylcholinesterase inhibitory activity. Phytotherapy Research, 21(12): 1142–1145. doi: 10.1002/ptr.2224 Nagamine T, Hayakawa K, Kusakabe T, et al. 2009. Inhibitory effect of fucoidan on Huh7 hepatoma cells through downregulation of CXCL12. Nutrition and Cancer, 61(3): 340–347. doi: 10.1080/01635580802567133 Natarajan S, Shanmugiahthevar K P, Kasi P D. 2009. Cholinesterase inhibitors from Sargassum and Gracilaria gracilis: seaweeds inhabiting South Indian coastal areas (Hare Island, Gulf of Mannar). Natural Product Research, 23(4): 355–369. doi: 10.1080/14786410802156036 Nauseef W M. 2014. Isolation of human neutrophils from venous blood. In: Quinn M T, DeLeo F R, eds. Neutrophil Methods and Protocols. Totowa: Humana Press, 13–18 Palanisamy S, Vinosha M, Marudhupandi T, et al. 2017. Isolation of fucoidan from Sargassum polycystum brown algae: structural characterization, in vitro antioxidant and anticancer activity. International Journal of Biological Macromolecules, 102: 405–412. doi: 10.1016/j.ijbiomac.2017.03.182 Park H Y, Choi I W, Kim G Y, et al. 2015. Fucoidan induces G1 arrest of the cell cycle in EJ human bladder cancer cells through down-regulation of pRB phosphorylation. Revista Brasileira de Farmacognosia, 25(3): 246–251. doi: 10.1016/j.bjp.2015.03.011 Park H S, Hwang H J, Kim G Y, et al. 2013. Induction of apoptosis by fucoidan in human leukemia U937 cells through activation of p38 MAPK and modulation of Bcl-2 family. Marine Drugs, 11(7): 2347–2364. doi: 10.3390/md11072347 Ratcliffe N A, Mello C B, Garcia E S, et al. 2011. Insect natural products and processes: new treatments for human disease. Insect Biochemistry and Molecular Biology, 41(10): 747–769. doi: 10.1016/j.ibmb.2011.05.007 Redner R L. 2002. Variations on a theme: the alternate translocations in APL. Leukemia, 16(10): 1927–1932. doi: 10.1038/sj.leu.2402720 Saitoh Y, Nagai Y, Miwa N. 2009. Fucoidan-Vitamin C complex suppresses tumor invasion through the basement membrane, with scarce injuries to normal or tumor cells, via decreases in oxidative stress and matrix metalloproteinases. International Journal of Oncology, 35(5): 1183–1189 Sellimi S, Kadri N, Barragan-Montero V, et al. 2014. Fucans from a Tunisian brown seaweed Cystoseira barbata: structural characteristics and antioxidant activity. International Journal of Biological Macromolecules, 66: 281–288. doi: 10.1016/j.ijbiomac.2014.02.041 Shen Zhixiang, Shi Zhanzhong, Fang Jing, et al. 2004. All-trans retinoic acid/As2O3 combination yields a high quality remission and survival in newly diagnosed acute promyelocytic leukemia. Proceedings of the National Academy of Sciences of the United States of America, 101(15): 5328–5335. doi: 10.1073/pnas.0400053101 Sobin L H, Fleming I D. 1997. TNM classification of malignant tumors, fifth edition (1997). Union internationale contre le cancer and the American joint committee on cancer. Cancer, 80(9): 1803–1804. doi: 10.1002/(SICI)1097-0142(19971101)80:9<1803::AID-CNCR16>3.0.CO;2-9 Sohrabipour J, Rabiei R. 1999. A list of marine algae of seashores of Persian Gulf and Oman Sea in the Hormozgan Province. Iranian Journal of Botany, 8(1): 131–162 Sohrabipour J, Rabiei R. 2007. The checklist of green algae of the Iranian coastal lines of the Persian Gulf and Gulf of Oman. The Iranian Journal of Botany, 13(2): 146–149 Vaziri Zadeh A, Mohammadi M, Fakhri A. 2012. Ecological assessment of mollusc communities in the rocky shores of Bushehr Province. Journal of Oceanography, 3(9): 55–61 Veena C K, Josephine A, Preetha S, et al. 2007. Effect of sulphated polysaccharides on erythrocyte changes due to oxidative and nitrosative stress in experimental hyperoxaluria. Human & Experimental Toxicology, 26(12): 923–932 Wang Yu, Xing Maochen, Cao Qi, et al. 2019. Biological activities of fucoidan and the factors mediating its therapeutic effects: a review of recent studies. Marine Drugs, 17(3): 183. doi: 10.3390/md17030183 Warrell Jr R P, Frankel S R, Miller Jr W H, et al. 1991. Differentiation therapy of acute promyelocytic leukemia with tretinoin (all-trans-retinoic acid). New England Journal of Medicine, 324(20): 1385–1393. doi: 10.1056/NEJM199105163242002 Warrell Jr R P, Maslak P, Eardley A, et al. 1994. Treatment of acute promyelocytic leukemia with all-trans retinoic acid: an update of the New York experience. Leukemia, 8(6): 929–933 Wei Chunmei, Xiao Qing, Kuang Xingyi, et al. 2015. Fucoidan inhibits proliferation of the SKM-1 acute myeloid leukaemia cell line via the activation of apoptotic pathways and production of reactive oxygen species. Molecular Medicine Reports, 12(5): 6649–6655. doi: 10.3892/mmr.2015.4252 Wong K K, Lawrie C H, Green T M. 2019. Oncogenic roles and inhibitors of DNMT1, DNMT3A, and DNMT3B in acute myeloid leukaemia. Biomarker Insights, 14: 1–12. doi: 10.1177/1177271919846454 Yamaguchi T, Takamura H, Matoba T, et al. 1998. HPLC method for evaluation of the free radical-scavenging activity of foods by using 1, 1-diphenyl-2-picrylhydrazyl. Bioscience, Biotechnology, and Biochemistry, 62(6): 1201–1204 Yan Mingde, Yao C J, Chow J M, et al. 2015. Fucoidan elevates microRNA-29b to regulate DNMT3B-MTSS1 axis and inhibit EMT in human hepatocellular carcinoma cells. Marine Drugs, 13(10): 6099–6116. doi: 10.3390/md13106099 Yang Meixiang, Ma Chunhong, Sun Jintang, et al. 2008. Fucoidan stimulation induces a functional maturation of human monocyte-derived dendritic cells. International Immunopharmacology, 8(13–14): 1754–1760 Yende S R, Harle U N, Chaugule B B. 2014. Therapeutic potential and health benefits of Sargassum species. Pharmacognosy Review, 8(15): 1–7. doi: 10.4103/0973-7847.125514 Yuan Xiumei, Zeng Yawei, Nie Kaiying, et al. 2015. Extraction optimization, characterization and bioactivities of a major polysaccharide from Sargassum thunbergii. PLoS ONE, 10(12): e0144773. doi: 10.1371/journal.pone.0144773 Zhang Zhongyuan, Teruya K, Eto H, et al. 2011. Fucoidan extract induces apoptosis in MCF-7 cells via a mechanism involving the ROS-dependent JNK activation and mitochondria-mediated pathways. PLoS ONE, 6(11): e27441. doi: 10.1371/journal.pone.0027441 Zhang Qingyu, Wang Feixuan, Jia Keke, et al. 2018. Natural product interventions for chemotherapy and radiotherapy-induced side effects. Frontiers in Pharmacology, 9: 1253. doi: 10.3389/fphar.2018.01253 -



 下载:
下载: