Comparison of short-term toxicity of 14 common phycotoxins (alone and in combination) to the survival of brine shrimp Artemia salina
-
Abstract: Toxic harmful algal blooms (HABs) can cause deleterious effects in marine organisms, threatening the stability of marine ecosystems. It is well known that different strains, natural populations and growth conditions of the same toxic algal species may lead to different amount of phycotoxin production and the ensuing toxicity. To fully assess the ecological risk of toxic HABs, it is of great importance to investigate the toxic effects of phycotoxins in marine organisms. In this study, the short-term toxicity of 14 common phycotoxins (alone and in combination) in the marine zooplankton Artemia salina was investigated. The 48 h LC50 of the 14 phycotoxins varied from 0.019 3 µg/mL to 2.415 µg/mL. The most potent phycotoxin was azaspiracids-3 (AZA3; with a LC50 of 0.019 3 µg/mL), followed by azaspiracids-2 (AZA2; 0.022 6 µg/mL), pectenotoxin-2 (PTX2; 0.046 0 µg/mL) and dinophysistoxin-1 (DTX1; 0.081 8 µg/mL). For the binary exposure, okadaic acid (OA) induced potential additive effects with DTX1, probably due to their similar structure (polyether fatty acid) and mode of action (attacking the serine/threonine phosphoprotein phosphatases). On the other hand, OA showed potential antagonistic effects with PTX2, which might be accounted for by their activation on the detoxification activity of cytochrome P450 activity. In addition, DTX1 induced potential synergetic effects with saxitoxin (STX), yessotoxin (YTX) or PTX2, suggesting the hazard potency of the mixtures of DTX1 and other phycotoxins (like STX, YTX and PTX2) with regard to the ecological risk. These results provide valuable toxicological data for assessing the impact of phycotoxins on marine planktonic species and highlight the potential ecological risk of toxic HABs in marine ecosystems.
-
Key words:
- LC50 /
- harmful algal blooms /
- binary exposure /
- ecological risk
-
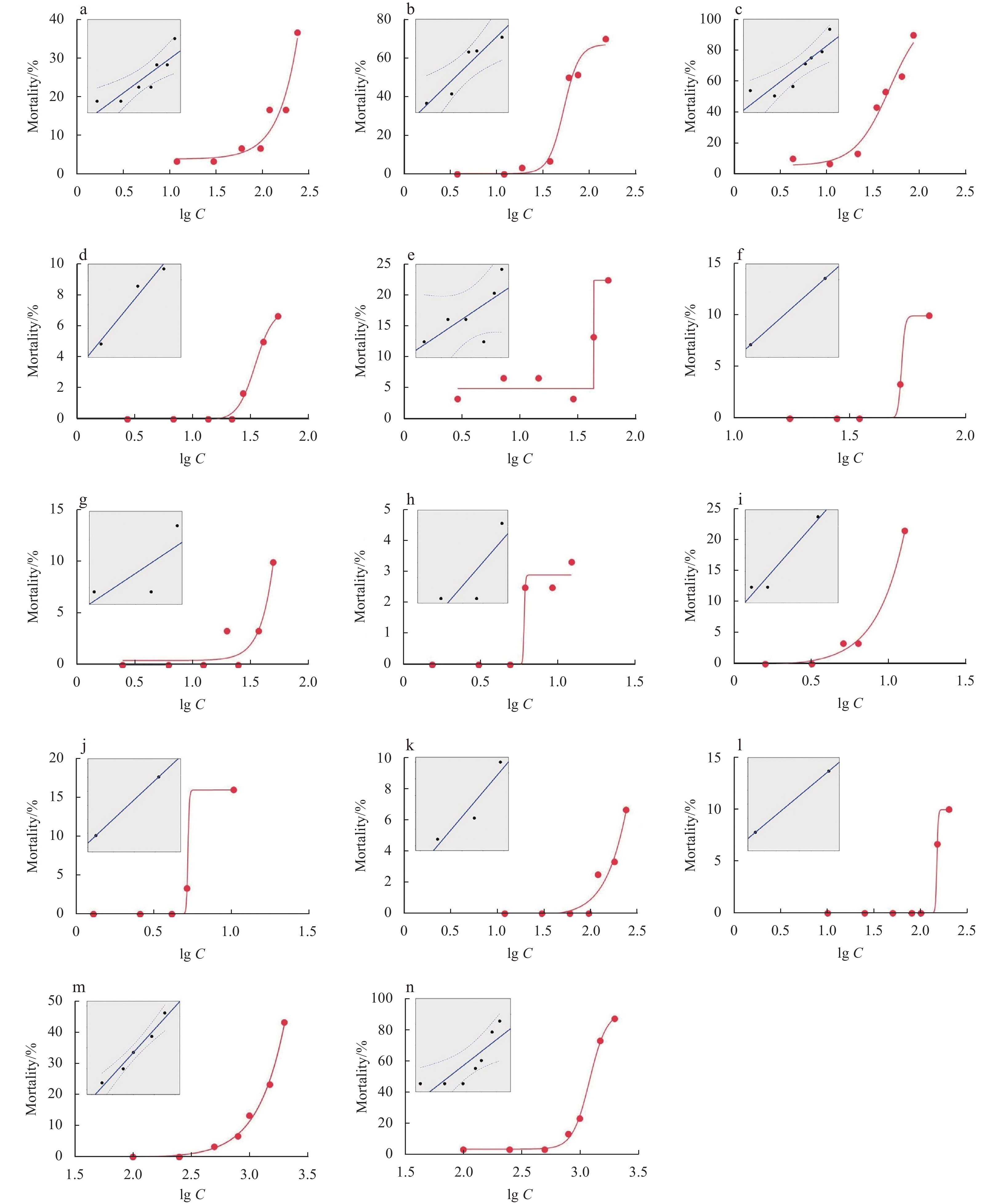
Figure 1. The 48 h mortality-concentration curves of OA (a), DTX1 (b), PTX2 (c), YTX (d), hYTX (e), SPX1 (f), GYM (g), AZA1 (h), AZA2 (i), AZA3 (j), STX (k), dcSTX (l), PbTx2 (m) and PbTx3 (n) in Artemia salina, where Y−axis is mortality and X−axis is the log10 concentration (unit: ng/mL) of phycotoxins (lg C). The insets are plots of probit transformed responses, where the Y−axis is probit and the X−axis is lg C.
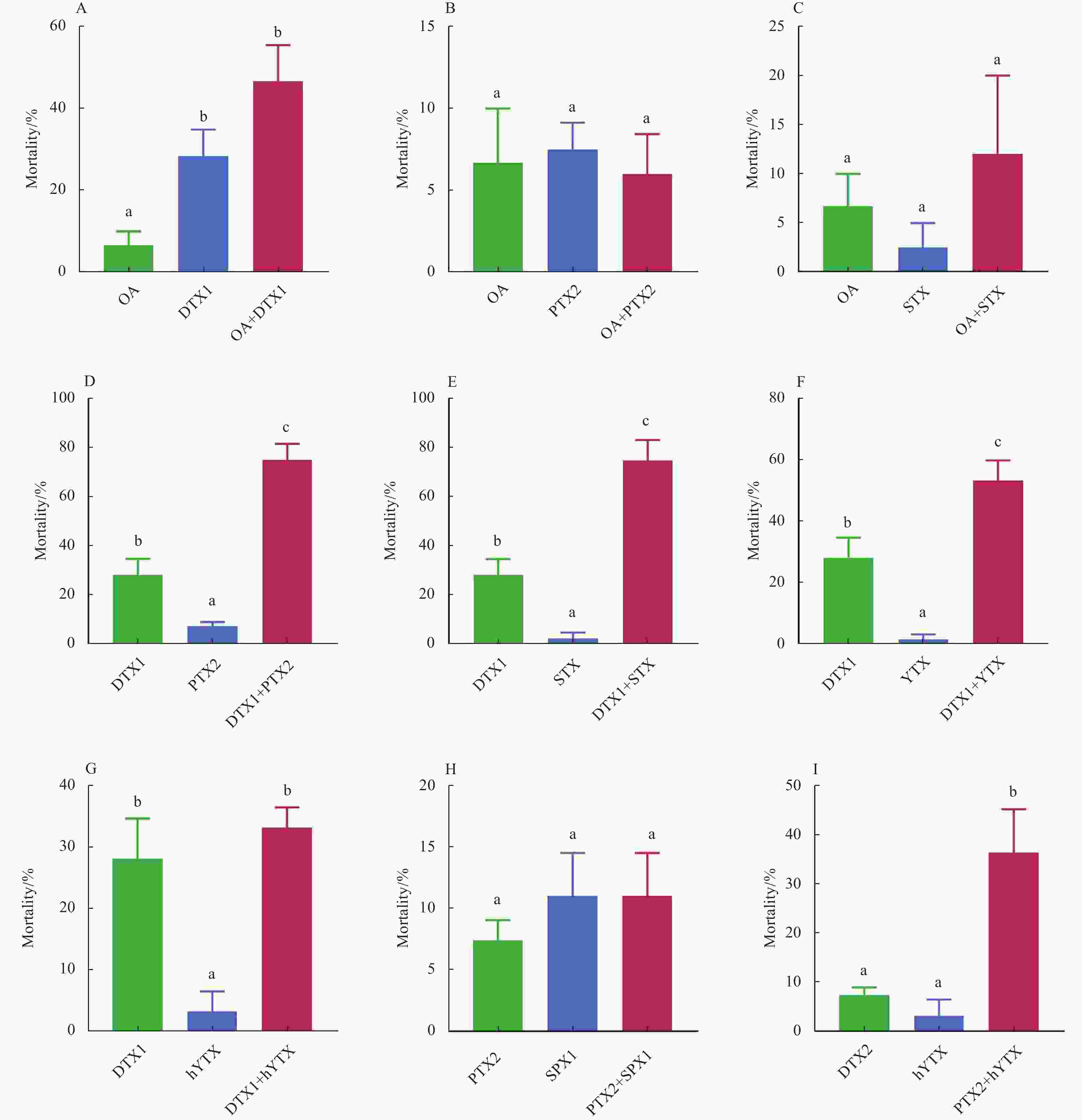
Figure 2. The individual and combined effects of OA+DTX1 (A), OA+PTX2 (B), OA+STX (C), DTX1+PTX2 (D), DTX1+STX (E), DTX1+YTX (F), DTX1+hYTX (G), PTX2+SPX1 (H) and PTX2+hYTX (I) on the 48 h mortality of Artemia salina. Data were expressed as the mean±SEM (n>3). Bars marked with different letters are significantly different from each other (p<0.05).
Table 1. The 48 h LC50 and LC10 values of marine phycotoxins in Artemia salina (n=3)
Phycotoxins LC50/(µg·mL−1) LC10/(µg·mL−1) OA 0.372 [0.287–0.746] 0.124 [0.074 8–0.153] DTX1 0.081 8 [0.046 0–0.139] 0.029 9 [0.003 27–0.050 7] PTX2 0.046 0 [0.035 2–0.057 3] 0.021 2 [0.009 57–0.029 4] YTX 0.171 [0.097 5–0.208] 0.061 2 [0.039 7–0.133] hYTX 0.085 9 [0.068 9–0.237] 0.048 0 [0.035 9–0.054 8] GYM 0.191 [0.102–1.667] 0.054 5 [0.042 7–0.102] SPX1 0.118 [0.091 0–0.345] 0.069 2 [0.061 7–0.087 9] AZA1 0.106 [0.032 4–105.44] 0.021 9 [0.013 2–5.701] AZA2 0.022 6 [0.017 2–0.038 5] 0.008 89 [0.007 53–0.010 5] AZA3 0.019 3 [0.014 5–0.036 8] 0.008 55 [0.007 24–0.010 3] STX 0.899 [0.469–16.520] 0.288 [0.222–0.716] dcSTX 0.376 [0.281–0.962] 0.194 [0.171–0.242] PbTx2 2.415 [2.056–3.499] 0.893 [0.423–1.161] PbTx3 1.279 [1.208–1.355] 0.811 [0.719–0.887] Note: The 95% confidence interval are given in brackets. Table 2. List of the recent toxicological data about the toxicity of phycotoxins in aquatic organisms
Phycotoxin Species Time LC50/(µg·mL−1) Reference OA Tigriopus californicus 24 h 41.7 Shaw et al. (1997) Artemia franciscana 24 h 6 270* D’ors et al. (2014) Danio rerio larvae 24 h 10 Figueroa et al. (2020) Danio rerio larvae 48 h 8.5 Figueroa et al. (2020) Danio rerio larvae 72 h 7 Figueroa et al. (2020) Daphnia magna 48 h 42.1 Rambla-Alegre et al. (2018) Daphnia magna 96 h 0.003 Rambla-Alegre et al. (2018) Artemia salina 48 h 0.728 this study DTX1 Danio rerio larvae 24 h 7 Figueroa et al. (2020) Danio rerio larvae 48 h 5.5 Figueroa et al. (2020) Danio rerio larvae 72 h 5 Figueroa et al. (2020) Daphnia magna 48 h 29 Rambla-Alegre et al. (2018) Daphnia magna 96 h 0.008 Rambla-Alegre et al. (2018) Artemia salina 48 h 0.081 9 this study STX Artemia franciscana 24 h 4 060* D’ors et al. (2014) Artemia salina 48 h 1.042 32 this study PbTx Bambusia affinis 24 h 0.000 011 Kirkpatrick et al. (2004) Oryzias latipes 24 h 0.015–25 Poli (1988) PbTx2 Artemia salina 48 h 2.415 this study PbTx3 Artemia salina 48 h 1.239 this study Note: * represents the calculated equivalent. -
Abdenadher M, Hamza A, Fekih W, et al. 2012. Factors determining the dynamics of toxic blooms of Alexandrium minutum during a 10-year study along the shallow southwestern Mediterranean coasts. Estuarine, Coastal and Shelf Science, 106: 102–111 Alarcan J, Barbé S, Kopp B, et al. 2019. Combined effects of okadaic acid and pectenotoxin-2, 13-desmethylspirolide C or yessotoxin in human intestinal Caco-2 cells. Chemosphere, 228: 139–148. doi: 10.1016/j.chemosphere.2019.04.018 Alarcan J, Biré R, Le Hégarat L, et al. 2018. Mixtures of lipophilic phycotoxins: exposure data and toxicological assessment. Marine Drugs, 16(2): 46. doi: 10.3390/md16020046 Alarcan J, Dubreil E, Huguet A, et al. 2017. Metabolism of the marine phycotoxin PTX-2 and its effects on hepatic xenobiotic metabolism: activation of nuclear receptors and modulation of the phase I cytochrome P450. Toxins, 9(7): 212. doi: 10.3390/toxins9070212 Alfonso C, Rehmann N, Hess P, et al. 2008. Evaluation of various pH and temperature conditions on the stability of azaspiracids and their importance in preparative isolation and toxicological studies. Analytical Chemistry, 80(24): 9672–9680. doi: 10.1021/ac801506d Anderson D M, Alpermann T J, Cembella A D, et al. 2012. The globally distributed genus Alexandrium: multifaceted roles in marine ecosystems and impacts on human health. Harmful Algae, 14: 10–35. doi: 10.1016/j.hal.2011.10.012 Bravo I, Fernández M L, Ramilo I, et al. 2001. Toxin composition of the toxic dinoflagellate Prorocentrum lima isolated from different locations along the Galician Coast (NW Spain). Toxicon, 39(10): 1537–1545. doi: 10.1016/S0041-0101(01)00126-X Chen Junhui, Han Tongzhu, Li Xiaotong, et al. 2018. Occurrence and distribution of marine natural organic pollutants: Lipophilic marine algal toxins in the Yellow Sea and the Bohai Sea, China. Science of the Total Environment, 612: 931–939. doi: 10.1016/j.scitotenv.2017.08.304 De Rijcke M, Van Acker E, Nevejan N, et al. 2016. Toxic dinoflagellates and Vibrio spp. act independently in bivalve larvae. Fish & Shellfish Immunology, 57: 236–242 Dickey R W, Bobzin S C, Faulkner D J, et al. 1990. Identification of okadaic acid from a Caribbean dinoflagellate, Prorocentrum concavum. Toxicon, 28(4): 371–377. doi: 10.1016/0041-0101(90)90074-H Donovan C J, Ku J C, Quilliam M A, et al. 2008. Bacterial degradation of paralytic shellfish toxins. Toxicon, 52(1): 91–100. doi: 10.1016/j.toxicon.2008.05.005 D’ors A, Bartolomé M C, Sánchez-Fortún S. 2014. Risk associated with toxic blooms of marine phytoplankton functional groups on Artemia franciscana. Journal of Coastal Life Medicine, 2(8): 625–631 Durbin E, Teegarden G, Campbell R, et al. 2002. North Atlantic right whales, Eubalaena glacialis, exposed to paralytic shellfish poisoning (PSP) toxins via a zooplankton vector, Calanus finmarchicus. Harmful Algae, 1(3): 243–251. doi: 10.1016/S1568-9883(02)00046-X Eckford-Soper L K, Daugbjerg N. 2017. Interspecific competition study between Pseudochattonella farcimen and P. verruculosa (Dictyochophyceae)—Two ichthyotoxic species that co-occur in Scandinavian waters. Microbial Ecology, 73(2): 259–270. doi: 10.1007/s00248-016-0856-z EFSA Panel on Contaminants in the Food Chain (CONTAM). 2010. Scientific Opinion on marine biotoxins in shellfish–Cyclic imines (spirolides, gymnodimines, pinnatoxins and pteriatoxins). EFSA Journal, 8(6): 1628 Faassen E J, Harkema L, Begeman L, et al. 2012. First report of (homo)anatoxin-a and dog neurotoxicosis after ingestion of benthic cyanobacteria in The Netherlands. Toxicon, 60(3): 378–384. doi: 10.1016/j.toxicon.2012.04.335 Farabegoli F, Blanco L, Rodríguez L P, et al. 2018. Phycotoxins in marine shellfish: origin, occurrence and effects on humans. Marine Drugs, 16(6): 188. doi: 10.3390/md16060188 Ferreiro S F, Vilariño N, Carrera C, et al. 2016. Subacute cardiovascular toxicity of the marine phycotoxin azaspiracid-1 in rats. Toxicological Sciences, 151(1): 104–114. doi: 10.1093/toxsci/kfw025 Ferron P J, Dumazeau K, Beaulieu J F, et al. 2016a. Combined effects of lipophilic phycotoxins (okadaic acid, azapsiracid-1 and yessotoxin) on human intestinal cells models. Toxins, 8(2): 50. doi: 10.3390/toxins8020050 Ferron P J, Hogeveen K, De Sousa G, et al. 2016b. Modulation of CYP3A4 activity alters the cytotoxicity of lipophilic phycotoxins in human hepatic HepaRG cells. Toxicology in Vitro, 33: 136–146. doi: 10.1016/j.tiv.2016.02.021 Fidler A E, Holland P T, Reschly E J, et al. 2012. Activation of a tunicate (Ciona intestinalis) xenobiotic receptor orthologue by both natural toxins and synthetic toxicants. Toxicon, 59(2): 365–372. doi: 10.1016/j.toxicon.2011.12.008 Figueroa D, Signore A, Araneda O, et al. 2020. Toxicity and differential oxidative stress effects on zebrafish larvae following exposure to toxins from the okadaic acid group. Journal of Toxicology and Environmental Health, Part A, 83(15–16): 573–588 Hallegraeff G M. 1993. A review of harmful algal blooms and their apparent global increase. Phycologia, 32(2): 79–99. doi: 10.2216/i0031-8884-32-2-79.1 Hisem D, Hrouzek P, Tomek P, et al. 2011. Cyanobacterial cytotoxicity versus toxicity to brine shrimp Artemia salina. Toxicon, 57(1): 76–83. doi: 10.1016/j.toxicon.2010.10.002 Höglander H, Larsson U, Hajdu S. 2004. Vertical distribution and settling of spring phytoplankton in the offshore NW Baltic Sea proper. Marine Ecology Progress Series, 283: 15–27. doi: 10.3354/meps283015 Jones G J, Negri A P. 1997. Persistence and degradation of cyanobacterial paralytic shellfish poisons (PSPs) in freshwaters. Water Research, 31(3): 525–533. doi: 10.1016/S0043-1354(96)00134-0 Jonsson P R, Pavia H, Toth G. 2009. Formation of harmful algal blooms cannot be explained by allelopathic interactions. Proceedings of the National Academy of Sciences of the United States of America, 106(27): 11177–11182. doi: 10.1073/pnas.0900964106 Kirkpatrick B, Fleming L E, Squicciarini D, et al. 2004. Literature review of Florida red tide: implications for human health effects. Harmful Algae, 3(2): 99–115. doi: 10.1016/j.hal.2003.08.005 Lincoln R A, Strupinski K, Walker J M. 1996. The use of Artemia nauplii (brine shrimp larvae) to detect toxic compounds from microalgal cultures. International Journal of Pharmacognosy, 34(5): 384–389. doi: 10.1076/phbi.34.5.384.13255 Marrouchi R, Rome G, Kharrat R, et al. 2013. Analysis of the action of gymnodimine-A and 13-desmethyl spirolide C on the mouse neuromuscular system in vivo. Toxicon, 75: 27–34. doi: 10.1016/j.toxicon.2013.08.050 Pan Lei, Chen Junhui, He Xiuping, et al. 2020. Aqueous photodegradation of okadaic acid and dinophysistoxin-1: persistence, kinetics, photoproducts, pathways, and toxicity evaluation. Science of the Total Environment, 743: 140593. doi: 10.1016/j.scitotenv.2020.140593 Poli M A. 1988. Laboratory procedures for detoxification of equipment and waste contaminated with brevetoxins PbTx-2 and PbTx-3. Journal of Association of Official Analytical Chemists, 71(5): 1000–1002 Quilliam M A. 1999. Phycotoxins. Journal of AOAC International, 82(2): 495 Rambla-Alegre M, Miles C O, de la Iglesia P, et al. 2018. Occurrence of cyclic imines in European commercial seafood and consumers risk assessment. Environmental Research, 161: 392–398. doi: 10.1016/j.envres.2017.11.028 Rossini G P, Hess P. 2010. Phycotoxins: chemistry, mechanisms of action and shellfish poisoning. In: Luch A, ed. Molecular, Clinical and Environmental Toxicology: Volume 2: Clinical Toxicology. Basel: Birkhä user, 65–122 Sala G L, Bellocci M, Callegari F, et al. 2013. Azaspiracid-1 inhibits the maturation of cathepsin D in mammalian cells. Chemical Research in Toxicology, 26(3): 444–455. doi: 10.1021/tx300511z Shaw B A, Andersen R J, Harrison P J. 1997. Feeding deterrent and toxicity effects of apo-fucoxanthinoids and phycotoxins on a marine copepod (Tigriopus californicus). Marine Biology, 128(2): 273–280. doi: 10.1007/s002270050092 Simões E, Vieira R C, Schramm M A, et al. 2015. Impact of harmful algal blooms (Dinophysis acuminata) on the immune system of oysters and mussels from Santa Catarina, Brazil. Journal of the Marine Biological Association of the United Kingdom, 95(4): 773–781. doi: 10.1017/S0025315414001702 Smayda T J. 1997. What is a bloom? A commentary. Limnology and Oceanography, 42(5part2): 1132–1136. doi: 10.4319/lo.1997.42.5_part_2.1132 Spector I, Braet F, Shochet N R, et al. 1999. New anti-actin drugs in the study of the organization and function of the actin cytoskeleton. Microscopy Research and Technique, 47(1): 18–37. doi: 10.1002/(SICI)1097-0029(19991001)47:1<18::AID-JEMT3>3.0.CO;2-E Suganuma M, Fujiki H, Suguri H, et al. 1988. Okadaic acid: an additional non-phorbol-12-tetradecanoate-13-acetate-type tumor promoter. Proceedings of the National Academy of Sciences of the United States of America, 85(6): 1768–1771. doi: 10.1073/pnas.85.6.1768 Terao K, Ito E, Yanagi T, et al. 1986. Histopathological studies on experimental marine toxin poisoning. I. Ultrastructural changes in the small intestine and liver of suckling mice induced by dinophysistoxin-1 and pectenotoxin-1. Toxicon, 24(11–12): 1141–1151 Toyofuku H. 2006. Joint FAO/WHO/IOC activities to provide scientific advice on marine biotoxins (research report). Marine Pollution Bulletin, 52(12): 1735–1745. doi: 10.1016/j.marpolbul.2006.07.007 Turki S, Dhib A, Fertouna-Bellakhal M, et al. 2014. Harmful algal blooms (HABs) associated with phycotoxins in shellfish: what can be learned from five years of monitoring in Bizerte Lagoon (southern Mediterranean Sea)?. Ecological Engineering, 67: 39–47 Turner J T. 2014. Planktonic marine copepods and harmful algae. Harmful Algae, 32: 81–93. doi: 10.1016/j.hal.2013.12.001 Twiner M J, Hess P, Dechraoui M Y B, et al. 2005. Cytotoxic and cytoskeletal effects of azaspiracid-1 on mammalian cell lines. Toxicon, 45(7): 891–900. doi: 10.1016/j.toxicon.2005.02.015 Twiner M J, Rehmann N, Hess P, et al. 2008. Azaspiracid shellfish poisoning: a review on the chemistry, ecology, and toxicology with an emphasis on human health impacts. Marine Drugs, 6(2): 39–72. doi: 10.3390/md6020039 Vale C, Gómez-Limia B, Nicolaou K C, et al. 2007. The c-Jun-N-terminal kinase is involved in the neurotoxic effect of azaspiracid-1. Cellular Physiology and Biochemistry, 20(6): 957–966. doi: 10.1159/000110456 Wang Yueming, Ong S S, Chai S C, et al. 2012. Role of CAR and PXR in xenobiotic sensing and metabolism. Expert Opinion on Drug Metabolism & Toxicology, 8(7): 803–817 Xiao Xi, Agustí S, Pan Yaoru, et al. 2019. Warming amplifies the frequency of harmful algal blooms with eutrophication in Chinese coastal waters. Environmental Science & Technology, 53(22): 13031–13041 Xu Jiayi, Hansen P J, Nielsen L T, et al. 2017. Distinctly different behavioral responses of a copepod, Temora longicornis, to different strains of toxic dinoflagellates, Alexandrium spp. Harmful Algae, 62: 1–9. doi: 10.1016/j.hal.2016.11.020 Yasumoto T. 1990. Marine microorganisms toxins—an overview. In: Granéli E, Sundström B, Edler L, et al., eds. Toxic Marine Phytoplankton. New York: Elsevier, 3–8 Zhang Xue, Hu Hongying, Men Yujie, et al. 2009. Feeding characteristics of a golden alga (Poterioochromonas sp. ) grazing on toxic cyanobacterium Microcystis aeruginosa. Water Research, 43(12): 2953–2960. doi: 10.1016/j.watres.2009.04.003 -




 下载:
下载: